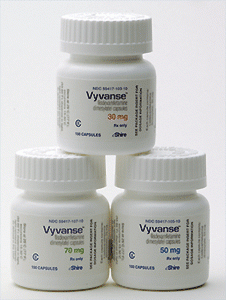
Buy Vyvanse 50mg online
$280.00 – $2,000.00
Molar mass: 263.378 g/mol
Bioavailability: 96.4%
Formula: C15H25N3O
Excretion: Renal: ~2%
Duration of action: 10–12 h
Onset of action: 2 h
Other names: (2S)-2,6-Diamino-N-, hexanamide; N–L-lysinamide
Guarantee Safe Checkout![]()
Description
Description
Has a generic version of Vyvanse been approved?
No. There is currently no therapeutically equivalent version of Vyvanse available in the United States.
Note: Fraudulent online pharmacies may attempt to sell an illegal generic version of Vyvanse. These medications may be counterfeit and potentially unsafe. If you purchase medications online, be sure you are buying from a reputable and valid online pharmacy. Ask your health care provider for advice if you are unsure about the online purchase of any medication.
Related patents
Patents are granted by the U.S. Patent and Trademark Office at any time during a drug’s development and may include a wide range of claims.
- Abuse-resistant amphetamine compounds
Patent 7,105,486
Issued: September 12, 2006
Inventor(s): Mickle; Travis & Krishnan; Suma & Bishop; Barney & Lauderback; Christopher & Moncrief; James Scott & Oberlender; Rob & Piccariello; Thomas
Assignee(s): New River Pharmaceuticals Inc.
The invention describes compounds, compositions and methods of using the same comprising a chemical moiety covalently attached to amphetamine. These compounds and compositions are useful for reducing or preventing abuse and overdose of amphetamine. These compounds and compositions find particular use in providing an abuse-resistant alternative treatment for certain disorders, such as attention deficit hyperactivity disorder (ADHD), ADD, narcolepsy, and obesity. Oral bioavailability of amphetamine is maintained at therapeutically useful doses. At higher doses bioavailability is substantially reduced, thereby providing a method of reducing oral abuse liability. Further, compounds and compositions of the invention decrease the bioavailability of amphetamine by parenteral routes, such as intravenous or intranasal administration, further limiting their abuse liability.Pediatric exclusivity - Abuse resistant lysine amphetamine compounds
Patent 7,223,735
Issued: May 29, 2007
Inventor(s): Mickle; Travis & Krishnan; Suma & Moncrief; James Scott & Lauderback; Christopher & Bishop; Barney & Oberlender; Rob & Piccariello; Thomas
Assignee(s): New River Pharmaceuticals Inc.
The present invention describes compounds, compositions and methods of using the same comprising lysine covalently attached to amphetamine. These compounds and compositions are useful for reducing or preventing abuse and overdose of amphetamine. These compounds and compositions find particular use in providing an abuse-resistant alternative treatment for certain disorders, such as attention deficit hyperactivity disorder (ADHD), ADD, narcolepsy, and obesity. Oral bioavailability of amphetamine is maintained at therapeutically useful doses. At higher doses bioavailability is substantially reduced, thereby providing a method of reducing oral abuse liability. Further, compounds and compositions of the invention decrease the bioavailability of amphetamine by parenteral routes, such as intravenous or intranasal administration, further limiting their abuse liability.
Additional information
Additional information
| Dosage | 30 MG, 40 MG, 50 MG, 70 MG |
|---|---|
| Quantity | 30 pills, 60 pills, 90 pills, 100 pills, 150 pills, 180 pills, 200 pills, 250 pills, 300 pills, 500 pills |




Michael (verified owner) –
Very fast delivery.
Camden (verified owner) –
Very well worth the money.
Matthew (verified owner) –
Good service.
Kayden (verified owner) –
Good quality.
Richard (verified owner) –
Very fast delivery.
Jackson (verified owner) –
Good quality.
Dominic (verified owner) –
Very well worth the money.
Lucas (verified owner) –
Good service.
Jackson (verified owner) –
Very well worth the money.
Paul (verified owner) –
Very fast delivery.